BOSENTIME 62.5
Item requires a valid prescription
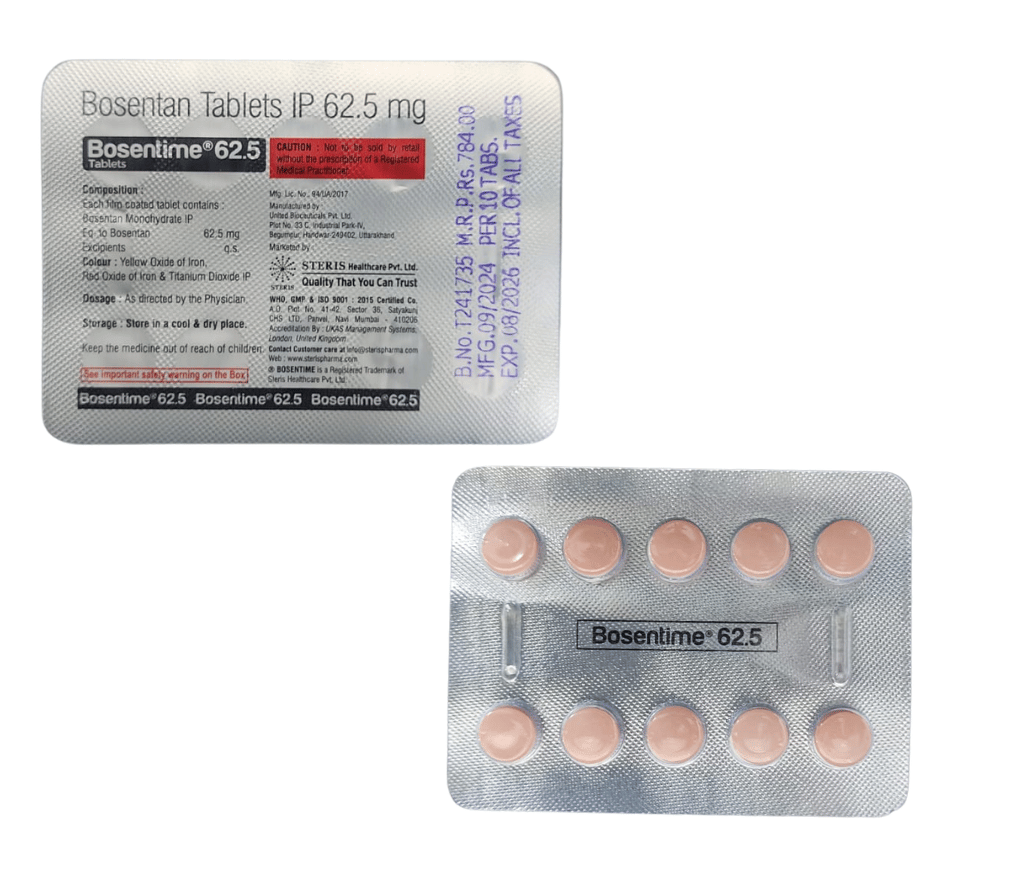
Manufactured By Steris Healthcare Pvt Ltd
Composition bosentan 62.5 mg
Rs 514.50
MRP Rs 735.00
(30% OFF)
Includes all taxes
Package SIZE
( For 10 Tablets )
100% Authentic
Products
Free
Shipping*
Products
Return Policy
Description:
BOSENTIME 62.5 is a prescription medication that contains Bosentan 62.5 mg, a potent endothelin receptor antagonist used primarily in the management of pulmonary arterial hypertension (PAH). Manufactured with stringent quality standards, BOSENTIME 62.5 by Steris Healthcare is designed to improve exercise capacity and slow disease progression in patients diagnosed with PAH. The medicine works by blocking the effects of endothelin-1, a naturally occurring substance in the body that causes blood vessels to narrow. By inhibiting this effect, Bosentan helps in dilating the blood vessels, thereby reducing blood pressure in the lungs and improving heart function.
What is BOSENTIME 62.5 (Bosentan 62.5 mg)?
BOSENTIME 62.5 is a tablet formulation of Bosentan, administered orally under a doctor’s supervision. It is indicated for patients suffering from pulmonary arterial hypertension, a condition characterized by high blood pressure in the arteries of the lungs. This condition leads to shortness of breath, dizziness, fatigue, and chest pain. BOSENTIME 62.5 improves the ability to exercise and delays clinical worsening of the disease.
Uses of BOSENTIME 62.5
-
Treatment of pulmonary arterial hypertension (PAH) in adults and children over 12 years of age.
-
Enhances exercise capacity in affected patients.
-
Slows the progression of symptoms such as shortness of breath and fatigue.
-
May also be used off-label in systemic sclerosis-related digital ulcers, depending on the physician’s discretion.
Side Effects of BOSENTIME 62.5
Like all medications, BOSENTIME 62.5 may cause side effects in some individuals. Common side effects include:
-
Headache
-
Respiratory tract infections
-
Ankle swelling (edema)
-
Flushing
-
Low red blood cell count (anemia)
-
Liver enzyme abnormalities
-
Nasal congestion
-
Rash or itching
Rare but serious side effects include liver damage, hypotension, or signs of allergic reactions such as difficulty breathing and facial swelling. Regular monitoring of liver function and blood count is advised during treatment.
Precautions Before Using BOSENTIME 62.5
-
Liver Monitoring: Bosentan can cause liver injury; liver function tests should be done prior to starting and regularly during treatment.
-
Pregnancy Warning: Bosentan is harmful to unborn babies. Effective contraception must be used during therapy.
-
Breastfeeding: Not recommended during treatment.
-
Anemia Risk: Monitor hemoglobin levels regularly.
-
Inform your doctor if you have a history of liver disease, heart failure, or low blood pressure.
Drug Interactions – What Drugs Interact with BOSENTIME 62.5?
BOSENTIME 62.5 can interact with several medications, which may increase side effects or reduce efficacy:
-
Cyclosporine A: Avoid combination due to increased bosentan levels.
-
Glyburide (Glibenclamide): Risk of elevated liver enzymes.
-
Hormonal Contraceptives: Bosentan may reduce effectiveness—use alternative or additional contraceptive methods.
-
Rifampin: May reduce bosentan blood levels, decreasing its effectiveness.
-
Other medications including ketoconazole, simvastatin, and warfarin may require dosage adjustments or closer monitoring.
Always inform your healthcare provider about all the medicines, supplements, and herbal products you are taking before starting BOSENTIME 62.5.
Conclusion
BOSENTIME 62.5 (Bosentan 62.5 mg) is a clinically effective option in the management of pulmonary arterial hypertension, offering improved symptom control and exercise tolerance. Its mechanism in blocking endothelin receptors helps relieve pressure in the lungs and enhances patient quality of life. While highly beneficial, it must be taken under medical supervision due to its potential for liver toxicity and other interactions. Choose BOSENTIME 62.5 from Steris Healthcare for assured quality, reliability, and results in PAH therapy.